The insertion of a central venous catheter (CVC) is a common procedure carried out in critically ill patients to administer fluids, medications, and assist in hemodynamic monitoring. CVC insertion possesses potential risks for patients both short term and long term (1). Some immediate mechanical complications are potentially life threatening such as pneumothorax, hemothorax, misplacement and arterial puncture with severe bleeding (1). In earlier studies the incidence of mechanical complications have been reported as high as 18.7% (2) and in newer studies as low as 0.1% (3).
Because of these potential risks most institutions mandate in their guidelines that immediate complications are ruled out by the routine use of a postprocedural chest x-ray (CXR). The routine use of a postprocedural CXR consumes valuable health care resources, is not risk-free for patients or staff as it adds to the cumulative cancer risk, and the specificity and sensitivity of CXR not as high as we would wish (1, 3-9). Furthermore, interpretation of CVC tip placement and pneumo- or hemo- thorax on CXR requires substantial training and is often not accurate even in experienced radiologists (10, 11).
The increasing and almost universal routine use of ultrasound (US) – guided CVC insertion have reduced both immediate and long term complications dramatically, reduced failure rates, procedure time, and improved especially novices’ first pass attempts, and is now widely considered the gold standard (4, 12-19).
This increasing use of US guidance to assist insertion has questioned the routine use of CXR as the gold standard for verification of tip placement and to rule out pneumo- and hemo-thorax. And several studies have looked into this specific use of bedside use of US at CVC insertion (3-8, 16, 20-24).
The additional potential benefits to further use of US to rule out complications after CVC insertion are less radiation to the patient, less time to safe use of the catheter, and reduction in healthcare costs since a CXR is expensive compared to the widely available bedside US machines.
Misplacement
Malpositioning of CVCs is more frequent than pneumothorax in the literature. The correct placement for CVC is considered with the tip in the Superior Vena Cava (SVC) – atrial junction. Often, the CVC is considered misplaced when it is either too proximal in the SVC or too deep in the right atrium or ventricle, or coiled or looped along its course. Less frequently is the displacement to the brachiocephalic veins. However the safest location of the CVC is still a topic of debate and evidence suggests that besides CVCs placed in the internal jugular vein CVCs with the tip in the RA, SVC, both brachiocephalic or subclavian veins are well tolerated (5, 6, 20, 25, 26).
In a recent Canadian study of 6875 CVCs, misplacement was reported in 1.91 % of the procedures and the study reported that rather than patient characteristics, the site of insertion had influence on the risk of misplacement, with the right subclavian veins most frequently leading to misplacement (5). In a systematic review and meta-analysis from 2018 of 2602 CVCs, misplacements were reported in 6.8% and a diagnostic accuracy of recognising misplacement with US had a specificity of 98.5% and a sensitivity of 68.2%. With the use of additional vascular US with Trans Thoracic Echocardiography (TTE) the sensitivity could be increased to 96.1% (6). Other advantages was that US was performed faster in alle cases and thereby reducing time to safe use and preventing exposure of radiation for both patients and staff (20).
Using CXR to confirm tip placement holds its own limitations to its accuracy even with trained radiologist and in the absence of symptoms CXR should not be considered a reliable diagnostic method (10, 25-28).
The use of US to verify tip placement with the bubble test has an excellent sensitivity of 100% and specificity of 94% in one study after limited training of residents (15). In a systematic review and meta-analysis from 2018 the pooled specificity of US to detect CVC misplacement was reported to be 98.9% and sensitivity at 68,2%. The sensitivity could be raised to 96.1% when TTE was combined with vascular US and due to the low a priori risk of malpositioning the study still recommends US over CXR in the verification of tip placement (20) .
Based on the available evidence, US could be favored over CXR in evaluation of CVC misplacement. However, for both CXR and US caution should be taken in certain situations where misplacement is more likely. CVCs are more often misplaced in taller people and males and in these cases a CXR can be considered and when the LSV is chosen as the site of insertion (7). There is no significant relation between BMI and the risk of malpositioning (7).
In conclusion, the definiton of catheter malpositioning should probably be reconsidered and a protocol for tip placement include both cardia US with saline flush and vascular US, especially when the LSV is the site of insertion (6, 20, 29).
Misplacement of the CVC is also not always a clinically relevant event, and the CVC does not always require repositioning. Often the fear has been cardiac perforation if positioned to deep in the right atrium. This is not found in the literature investigating newer and more flexible catheters (30). The risk of malignant arrhythmias is extremely rare and with monitoring of patients during insertions arrhythmias are often reported as non-malignant and self-limiting (1). In the Canadian study only 24 out of 131 patients required repositioning. None of the misplaced CVCs resulted in complications for the patients (5).
Evaluation of tip placement
To confirm the placement of the CVC in the SVC -atrial junction use the cardiac or abdominal probe. The probe should be placed in the subcostal view, just below the sternum and with the marker to the patient’s right shoulder. The probe should be placed in a flat almost horisontal angle to obtain the best view. Slide and manipulate the probe until you obtain a four chamber view of the heart. After you have obtained a four chamber view, an assistant can quickly flush 10ml of normal saline into the distal leg of the CVC and an atrial swirl will be seen as bubbles in a dense laminar flow in the RA within 2 seconds. No CVC tip should be visible in the RA. If the flow of bubbles is seen immediately after injection and is very turbulent, the tip may be placed too deep in the RA. If the bubbles are absent or appear after more then 2 seconds, the CVC should be considered misplaced. Additional vascular US with scanning of especially the internal jugular veins or CXR can be performed to verify location and guide decision making on weather to reposition or not (16, 20-24, 31).
In the video below you can watch a video demonstrating how to perform the “bubble test”.
Pneumothorax
Recent larger retrospective studies have reported pneumothoraces as rare (1, 4-7, 20). In the recent Swedish study of 10.949 CVC insertions mechanical complications occurred in only 1.1% of the cases and only 0.2% were considered severe, where severe ment pneumothorax or severe bleeding (1). These complication rates are from a study where only half the procedures were reported as performed under US guidance (1). In the Canadian of 6.845 patients the overall incidence of pneumothorax was 0.33% and even lower and only 0.29% when the IRJ vein was used (5).
In both studies, the risk of pneumothorax is reported as more likely to happen when the LSV is the site of insertion and when arterial puncture happens (1, 5).
The diagnostic accuracy of US has been reported as superior to CXR in multiple studies and a recent systematic review with meta-analysis confirms this statement (6). In this study of 1553 CVC insertions there were 12 pneumothoraces (1.1%) and all were identified using US versus only 10 identified on CXR. Both the sensitivity and specificity from this review is 100% (6).
Evaluation of pneumothorax
Before the procedure, scan the patient’s lungs using the same approach as below to rule out pneumothorax before the procedure. After the placement of the CVC use the linear US probe to assess for pneumothorax again. The probe should be placed over the second or third intercostal space in the mid- clavicular line. Then identify the hyperechoic rib-rims with the acoustic shadow below and the hyperechoic pleural line. As the patients breathes lung sliding should be present and is best described as pearls on a string or tiny little ants walking along the pleural line. Furthermore, look for the presence of B-lines, that look like shimmering comet tails radiating downwards. A single B-line is enough to rule-out pneumothorax and holds a true-negative of 100% (21, 32).
After that, shift to M-mode on the US machine to further rule out pneumothorax. Look for the seashore sign in stead of stratosphere sign. The seashore sign looks like a sandy beach with water above it, shown in the video below. The stratosphere sign has “sea” lines all over the US image.
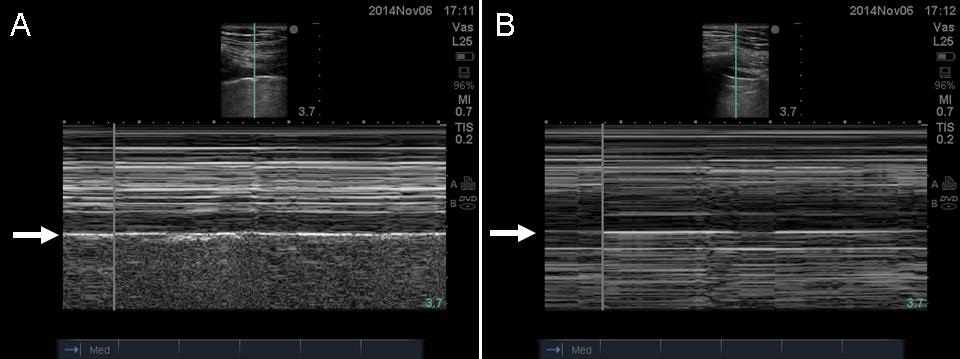
In the presence of pneumothorax there will be no lung sliding, no B-lines and instead of the sandy beach sign in the M-mode, you will find the stratosphere sign shown above in the picture marked as B.
If a pneumothorax is present, patients clinical status should guide treatment, not CXR or US findings.
Below you will find your very own little pocket guide to assist you in your daily (training of) US guided evaluation of pneumothorax and tip placement after US guided central venous catheter placement.

Barriers to use of US for evaluation of pneumothorax and confirmation of tip placement have been reported to be hospital policy and guideline, lack of US training and lack of confidence in US skills, lack of awareness that US could be used for postprocedual evaluation, operator convenience favoring CXR, and as always when it comes to implementation of new guidelines, a lack of interest in change in behaviour (33). Hopefully this blogpost can assist you in acquiring the knowledge needed to help you overcome the knowledge barrier, and hopefully the videos can help you understand “how to” US for postprocedural evaluation. The Pocket guide can assist you in your daily work at the bedside, reminding you to give it a go and assist you in performance. If you need additional knowledge about US lingo – read reference 32 (open access) and go find one of the early adopters in your department that can help you get started. Please remember that you should still follow hospital policy of postprocedural CXR while you practice your US skills.
Finally, in case you want to refine your CVC insertion technique even more, consider getting familiar with the oblique view, your microconvex probe and real time assessment of guidewire placement using vascular US. Robin Lundén from the scanfoam team and master mind behind InternetAnestesi (Swe) and InterAnest for english material has made this awesome #viking #FOAMed contribution in english for all of us to enjoy and learn from.
vb/ Sandra
References:
1.Björkander M, Bentzer P, Schött U, Broman ME, Kander T. Mechanical complications of central venous catheter insertions: A retrospective multicenter study of incidence and risks. Acta Anaesthesiol Scand. 2019 Jan;63(1):61-68.
2.Calvache JA, Rodríguez MV, Trochez A, Klimek M, Stolker RJ, Lesaffre E. Incidence of Mechanical Complications of Central Venous Catheterization Using Landmark Technique: Do Not Try More Than 3 Times. J Intensive Care Med. 2016 Jul;31(6):397-402.
3.Hourmozdi JJ, Markin A, Johnson B, Fleming PR, Miller JB. Routine Chest Radiography Is Not Necessary After Ultrasound-Guided Right Internal Jugular Vein Catheterization. Crit Care Med. 2016 Sep;44(9)
4.Molgaard O, Nielsen MS, Handberg BB, Jensen JM, Kjaergaard J, Juul N. Routine X-ray control of upper central venous lines: Is it necessary? Acta Anaesthesiol Scand. 2004 Jul;48(6):685-9.
5.Chui J, Saeed R, Jakobowski L, Wang W, Eldeyasty B, Zhu F, Fochesato L, Lavi R, Bainbridge D. Is Routine Chest X-Ray After Ultrasound-Guided Central Venous Catheter Insertion Choosing Wisely?: A Population-Based Retrospective Study of 6,875 Patients. Chest. 2018 Jul;154(1):148-156.
6.Ablordeppey EA, Drewry AM, Beyer AB, Theodoro DL, Fowler SA, Fuller BM, Carpenter CR. Diagnostic Accuracy of Central Venous Catheter Confirmation by Bedside Ultrasound Versus Chest Radiography in Critically Ill Patients: A Systematic Review and Meta-Analysis. Crit Care Med. 2017 Apr;45(4):715-724.
7.Woodland DC, Randall Cooper C, Farzan Rashid M, Rosario VL, Weyker PD, Weintraub J, Bentley-Hibbert S, Kluger MD. Routine chest X-ray is unnecessary after ultrasound-guided central venous line placement in the operating room. J Crit Care. 2018 Aug;46:13-16.
8.Bailey SH, Shapiro SB, Mone MC, Saffle JR, Morris SE, Barton RG. Is immediate chest radiograph necessary after central venous catheter placement in a surgical intensive care unit? Am J Surg. 2000 Dec;180(6):517-21; discussion 521-2.
9.Seleznova Y, Brass P, Hellmich M, Stock S, Müller D. Cost-effectiveness-analysis of ultrasound guidance for central venous catheterization compared with landmark method: a decision-analytic model. BMC Anesthesiol. 2019 Apr 9;19(1):51.
10.Abood GJ, Davis KA, Esposito TJ, Luchette FA, Gamelli RL. Comparison of routine chest radiograph versus clinician judgment to determine adequate central line placement in critically ill patients. J Trauma. 2007 Jul;63(1):50-6.
11.Chan TY, England A, Meredith SM, McWilliams RG. Radiologist variability in assessing the position of the cavoatrial junction on chest radiographs. Br J Radiol. 2016 Sep;89(1065):20150965.
12.Leung J, Duffy M, Finckh A. Real-time ultrasonographically-guided internal jugular vein catheterization in the emergency department increases success rates and reduces complications: a randomized, prospective study. Ann Emerg Med. 2006 Nov;48(5):540-7. Epub 2006 Feb 21.
13.Corvetto MA, Pedemonte JC, Varas D, Fuentes C, Altermatt FR. Simulation-based training program with deliberate practice for ultrasound-guided jugular central venous catheter placement. Acta Anaesthesiol Scand. 2017 Oct;61(9):1184-1191.
14.Griswold-Theodorson S, Hannan H, Handly N, Pugh B, Fojtik J, Saks M, Hamilton RJ, Wagner D. Improving patient safety with ultrasonography guidance during internal jugular central venous catheter placement by novice practitioners. Simul Healthc. 2009 Winter;4(4):212-6.
15.Frykholm P, Pikwer A, Hammarskjöld F, Larsson AT, Lindgren S, Lindwall R, Taxbro K, Oberg F, Acosta S, Akeson J. Clinical guidelines on central venous catheterisation. Swedish Society of Anaesthesiology and Intensive Care Medicine. Acta Anaesthesiol Scand. 2014 May;58(5):508-24.
16.Korsten P, Mavropoulou E, Wienbeck S, Ellenberger D, Patschan D, Zeisberg M, Vasko R, Tampe B, Müller GA. The “rapid atrial swirl sign” for assessing central venous catheters: Performance by medical residents after limited training. PLoS One. 2018 Jul 16;13(7):e0199345.
17.Brass P, Hellmich M, Kolodziej L, Schick G, Smith AF. Ultrasound guidance versus anatomical landmarks for subclavian or femoral vein catheterization. Cochrane Database Syst Rev. 2015 Jan 9;1:CD011447.
18.Saugel B, Scheeren TWL, Teboul JL. Ultrasound-guided central venous catheter placement: a structured review and recommendations for clinical practice. Crit Care. 2017 Aug 28;21(1):225.
19.McGee DC, Gould MK. Preventing complications of central venous catheterization. N Engl J Med. 2003 Mar 20;348(12):1123-33. Review.
20.Smit JM, Raadsen R, Blans MJ, Petjak M, Van de Ven PM, Tuinman PR. Bedside ultrasound to detect central venous catheter misplacement and associated iatrogenic complications: a systematic review and meta-analysis. Crit Care. 2018 Mar 13;22(1):65.
21.Saul T, Doctor M, Kaban NL, Avitabile NC, Siadecki SD, Lewiss RE. The Ultrasound-Only Central Venous Catheter Placement and Confirmation Procedure. J Ultrasound Med. 2015 Jul;34(7):1301-6.
22.Weekes AJ, Johnson DA, Keller SM, Efune B, Carey C, Rozario NL, Norton HJ. Central vascular catheter placement evaluation using saline flush and bedside echocardiography. Acad Emerg Med. 2014 Jan;21(1):65-72.
23.Duran-Gehring PE, Guirgis FW, McKee KC, Goggans S, Tran H, Kalynych CJ, Wears RL. The bubble study: ultrasound confirmation of central venous catheter placement. Am J Emerg Med. 2015 Mar;33(3):315-9.
24.Gekle R, Dubensky L, Haddad S, Bramante R, Cirilli A, Catlin T, Patel G, D’Amore J, Slesinger TL, Raio C, Modayil V, Nelson M. Saline Flush Test: Can Bedside Sonography Replace Conventional Radiography for Confirmation of Above-the-Diaphragm Central Venous Catheter Placement? J Ultrasound Med. 2015 Jul;34(7):1295-9.
25.Vesely TM. Central venous catheter tip position: a continuing controversy. J Vasc Interv Radiol. 2003 May;14(5):527-34.
26.Fletcher SJ, Bodenham AR. Safe placement of central venous catheters: where should the tip of the catheter lie? Br J Anaesth. 2000 Aug;85(2):188-91.
27.Dulce M, Steffen IG, Preuss A, Renz DM, Hamm B, Elgeti T. Topographic analysis and evaluation of anatomical landmarks for placement of central venous catheters based on conventional chest X-ray and computed tomography. Br J Anaesth. 2014 Feb;112(2):265-71.
28.Stonelake PA, Bodenham AR. The carina as a radiological landmark for central venous catheter tip position. Br J Anaesth. 2006 Mar;96(3):335-40.
29.Bedel J, Vallée F, Mari A, Riu B, Planquette B, Geeraerts T, Génestal M, Minville V, Fourcade O. Guidewire localization by transthoracic echocardiography during central venous catheter insertion: a periprocedural method to evaluate catheter placement. Intensive Care Med. 2013 Nov;39(11):1932-7.
30.Pittiruti M, Lamperti M. Late cardiac tamponade in adults secondary to tip position in the right atrium: an urban legend? A systematic review of the literature. J Cardiothorac Vasc Anesth. 2015 Apr;29(2):491-5.
31.Esmailian M, Azizkhani R, Najafi N. Saline Flush versus Chest x ray in Confirmation of Central Venous Catheter Placement; a Diagnostic Accuracy Study. Emerg (Tehran). 2017;5(1):e75.
32.Ultrasound: Basic understanding and learning the language. Ihnatsenka B, Boezaart AP. International Journal of Shoulder Surgery. 2010 Jul-Sep; 4(3): 55-62
33.Ablordeppey EA, Drewry AM, Theodoro DL, Tian L, Fuller BM, Griffey RT. Current Practices in Central Venous Catheter Position Confirmation by Point of Care Ultrasound: A Survey of Early Adopters. Shock. 2019 May;51(5):613-618.
Star skater, simulationista by day, anaesthesia by night and #meded choreographer. Coming to a SIM room near you. With a shark.

